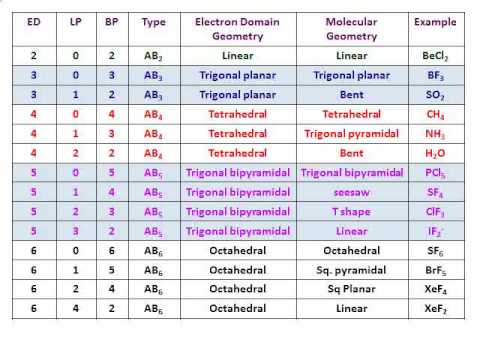
What is the relationship between geometric shape and electron pairs? Final Step Check The Stability Of Lewis Structure By Calculating The Formal Charge On Each Atom Thus, the electron– pair geometry is tetrahedral with three of the corners occupied by the bonding pairs of electrons. SOLUTION The Lewis structure for the SnCl3– ion is: The central Sn atom is surrounded by one nonbonding electron pair and three single bonds. Moreover, what is the electron pair geometry of sncl3? Electron domain geometries are one of the five learned so far: linear, trigonal planar, tetrahedral, trigonal bipyramidal, or octahedral. The total number of electron pairs, both bonding pairs and lone pairs, leads to what is called the electron domain geometry. While 3 electron domains means trigonal planar and 120 degrees.Īlso, what is the electron domain geometry? Central Atom with One or More Lone Pairs. 
For example, two electron domain would mean linear and 180 degree shape. Increasing electron domains, means changing name and shape. What Is The Connection Between The Number Of Electron Domains And The Electron Geometryģ.9/5 Electron geometry geometric ELECTRON number of electron Electron domain geometries number of electron bondingĪlso, how electronic shape is affected by the number of electron domains?Įlectronic shape and name is drastically influenced by the number of electrons domains. Choose a central atom: The Oxygen atom will be the central atom.Find the total number of bonds forming: Single covalent bonds between each oxygen and hydrogen atom.Look for how many electrons are needed: It is four for one water molecule according to the octet rule.Look for the total valence electrons: It is eight to form a single H2O molecule.Here, we need to understand how the Lewis structure is drawn for the H2O molecule: The Lewis structure of hydrogen and 2 oxygen atoms shows a total of eight valence electrons participate in the bond formation to form a single triatomic H2O molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
